A Complete Guide to Cellular Autophagy, Preconception Optimization, and Reproductive Health
Last Updated: March 2026 | Reading Time: ~45 minutes | Evidence Level: Peer-Reviewed Research
Table of Contents
- Introduction: The Preconception Revolution
- What Is Spermidine? The Biochemistry of Cellular Renewal
- Why Spermidine Levels Decline — And Why It Matters for Fertility
- Autophagy: The Cellular Foundation of Reproductive Health
- Spermidine and Female Fertility: Oocyte Quality and Ovarian Aging
- Spermidine and Male Fertility: Addressing the Silent 55%
- Oxidative Stress: The Common Enemy of Egg and Sperm Quality
- Bioavailability and Liposomal Spermidine
- The Autophagy-Optimized Conception Protocol (AOCP)
- Spermidine and IVF: The 1% Insurance Policy
- The Fasting-Mimicking Diet: Amplifying Autophagy Naturally
- Safety, Dosing, and Practical Implementation
- The Future of Fertility Science: What’s Coming Next
- Frequently Asked Questions (FAQ)
- Conclusion

Introduction: The Preconception Revolution
For decades, the fertility conversation followed a predictable script. That script was: take folic acid once pregnancy was confirmed, hope for the best, & address problems as they arose. Today, that script has been thrown out. A seismic shift is occurring in how educated, proactive couples approach family building. This shift is driven by necessity, empowered by emerging science, and fueled by the economic reality of assisted reproductive technology.
Women are delaying childbearing longer than ever before. First births to women over 35 have increased by 23% in the last decade alone. Male-factor infertility now contributes to roughly 55% of all infertility cases. Unfortunately, male reproductive health remains dramatically under-addressed in clinical settings. Meanwhile, a single IVF cycle costs $15,000–$30,000, with success rates that remain frustratingly modest — especially for women over 38. The emotional and financial toll is immense.
This climate has catalyzed an entirely new discipline: preconception optimization. Rather than reacting to infertility after it surfaces, proactive couples are now intervening months — sometimes years — before trying to conceive. They’re testing their ovarian reserve, analyzing sperm DNA fragmentation, and tracking mitochondrial health. Couples are now researching interventions that target the root cellular causes of reproductive aging. They’re asking not just “how do we get pregnant?” but “how do we produce the healthiest possible eggs and sperm?”
Preconception optimization treats fertility not as a lottery, but as a biological system that can be enhanced through cellular-level intervention. Prenatal vitamins are reactive — they support a pregnancy already established. Preconception supplementation is proactive — it targets the cellular foundations of conception itself.
Introducing Spermidine
At the center of this revolution is a molecule that most couples have never heard of, Spermidine. It may turn out to be one of the most important compounds for fertility optimization ever identified by modern science.
Spermidine is a naturally occurring polyamine found in every living cell. It was first identified in human semen in the 17th century — hence its name. Over the following two decades, science uncovered its profound connection to cellular longevity, autophagy, mitochondrial health, and fertility. Its levels decline significantly with age, tracking almost perfectly alongside the decline in both egg and sperm quality. When restored, it activates the very cellular repair systems that reproductive cells need to perform at their best.
My intent is to create this guide as the definitive resource on spermidine and fertility. We will explore the biochemistry of spermidine, the science of autophagy and mitophagy, landmark studies on oocyte and sperm quality. We’ll explain the critical importance of bioavailable liposomal delivery, and a complete Autophagy-Optimized Conception Protocol (AOCP) for couples. Every claim is grounded in peer-reviewed research, with direct links to studies so you can evaluate the evidence yourself.
Whether you are navigating advanced maternal age, supporting an IVF cycle, or optimizing reproductive health — this guide is for you.
What Is Spermidine? The Biochemistry of Cellular Renewal
Spermidine is a naturally occurring polyamine — a class of organic compounds defined by multiple amino groups. It is essential for cellular growth, proliferation, and differentiation. It belongs to the polyamine family, with which it shares a biosynthetic pathway catalyzed by the enzyme ornithine decarboxylase (ODC).
Spermidine was first isolated from human semen by the Dutch microscopist Anton van Leeuwenhoek in 1678. Spermidine was for centuries little more than a chemical curiosity with an unfortunate name. The last 30 years of molecular biology have transformed that entirely. We now understand spermidine as a master regulator of cellular health. It’s a molecule whose presence or absence dictates whether cells thrive, deteriorate, or die.
Core Biological Functions of Spermidine
Spermidine’s small molecular size is it’s advantage. It’s size & positive charge allow it to interact with the negatively charged phosphate backbone of DNA, RNA structures, and cell membranes. This gives it extraordinary reach within the cell:
- Gene expression regulation through chromatin remodeling and histone acetylation
- Protein synthesis support via translation initiation, ribosome assembly, and elongation factor activity
- DNA stability and repair — spermidine binds and protects DNA from oxidative damage
- Membrane integrity — stabilizes phospholipid bilayers and supports cellular signaling cascades
- Immune modulation — reduces pro-inflammatory cytokine production
- Mitochondrial biogenesis — promotes the creation of new, healthy mitochondria
- Autophagy induction — the mechanism most critical to fertility, discussed at length in Section 4
- Extension of lifespan — documented in model organisms from yeast (Saccharomyces cerevisiae) to nematodes (C. elegans) to flies (Drosophila) to mice
This breadth of function is unusual among dietary compounds. Which explains why spermidine research has exploded across disciplines including oncology, neuroscience, immunology, and — of course — reproductive biology.

Natural Dietary Sources of Spermidine
The body produces spermidine endogenously through the polyamine biosynthesis pathway, and also obtains it from dietary sources and gut microbiota. Foods naturally rich in spermidine include:
| Food Source | Spermidine Content (approx.) | Notes |
| Wheat germ | ~2,500 nmol/g | Highest known dietary source |
| Soybeans / soy products | ~900–1,900 nmol/g | Excellent daily source |
| Aged cheeses (cheddar, parmesan) | ~500–900 nmol/g | Convenient and concentrated |
| Mushrooms (shiitake, oyster) | ~700–900 nmol/g | Versatile culinary option |
| Chickpeas / lentils / legumes | ~400–700 nmol/g | Plant-based staple |
| Broccoli / cauliflower | ~200–400 nmol/g | Combine with other sources |
| Green peas | ~300–450 nmol/g | Easy addition to diet |
| Whole grains (oats, corn) | ~150–300 nmol/g | Broad dietary base |
| Certain fermented foods | Variable | Gut microbiome-dependent |
These dietary sources contribute meaningfully to total spermidine intake. However, diet alone is unlikely to achieve consistent, systemic levels shown in studies to meaningfully activate autophagy in reproductive tissues. This is especially important, particularly in the context of age-related decline. This is why supplementation, and specifically the form of supplementation, matters so profoundly for fertility applications (see below).
Spermidine’s Place in the Polyamine Family
Spermidine sits at the center of polyamine metabolism. It is synthesized from putrescine (via spermidine synthase) and can be converted to spermine (by spermine synthase). It can also be back-converted to putrescine through catabolic enzymes including spermidine/spermine N1-acetyltransferase (SSAT) and polyamine oxidase (PAO). This dynamic interconversion means that total polyamine homeostasis — the balance among putrescine, spermidine, and spermine — matters as much as absolute spermidine levels.
In fertility contexts, disruptions to this polyamine balance is directly linked to testicular dysfunction. It can also cause impaired folliculogenesis and reduced embryo viability. Restoring spermidine does not act in isolation — it rebalances a system that aging progressively disrupts.
Why Spermidine Levels Decline — And Why It Matters for Fertility
The most underappreciated fact about fertility is that spermidine levels in humans drop by up to 60% by middle age. This is not a minor fluctuation. It represents a fundamental shift in the cellular environment of every tissue in the body — including the ovaries and testes. The decline is driven by multiple converging factors:
- Reduced endogenous synthesis: Ornithine decarboxylase (ODC) activity decreases with age
- Increased catabolism: SSAT activity rises with age, accelerating spermidine breakdown
- Dietary pattern changes: Most Western diets are low in spermidine-rich whole foods
- Gut microbiome shifts: Age-related dysbiosis reduces microbially-produced polyamines
- Chronic inflammation: Inflammatory cytokines suppress polyamine biosynthetic pathways
The timeline of this decline maps almost perfectly onto the well-documented decline in human fertility. Women’s egg quality begins declining noticeably in the early 30s and accelerates dramatically after 35. Men’s sperm parameters — motility, morphology, DNA integrity — also show measurable decline with age, typically beginning in the mid-30s. The parallel is not coincidental.
The age-related decline in spermidine levels correlates disturbingly with the timeline of declining fertility in both men and women. This is not a peripheral association — it reflects a causal mechanism: when spermidine drops, autophagy slows, damaged cellular components accumulate, and the cellular environment of the ovaries and testes deteriorates.
Visualizing the Spermidine-Fertility Parallel

As shown in the infographic below, the decline in endogenous spermidine levels parallels reproductive aging almost exactly. This is why the Autophagy Optimized Conception Protocol emphasizes starting Liposomal Spermidine 3–6 months preconception — to activate mitophagy, clear damaged mitochondria, and combat oxidative stress before the steepest quality drops occur.
Effective for Men and Women Both
For women, this is particularly critical. A woman is born with all the eggs she will ever have — approximately 1–2 million primordial follicles at birth. However, this number declines to 300,000–400,000 by puberty, and continues to decrease throughout reproductive life. The quality of remaining eggs, not just their quantity, ultimately determines fertility outcomes. Egg quality is fundamentally a question of cellular health. Factors include integrity of mitochondria, chromosomal stability, efficiency of DNA repair, and competence of the cytoplasm for early embryonic development. All of these cellular quality parameters depend, in large part, on robust autophagy — and autophagy depends on spermidine.
For men, the dynamic is different but equally consequential. Unlike women, men produce sperm continuously throughout life, with a complete spermatogenic cycle taking approximately 74 days. This means any intervention that improves the cellular environment of sperm production can yield measurable benefits within roughly 2.5 months. However, sperm are uniquely vulnerable to oxidative stress and DNA damage due to their high metabolic activity. Other factors include minimal cytoplasm, high concentration of polyunsaturated fatty acids, and limited DNA repair mechanisms once fully formed.
Autophagy: The Cellular Foundation of Reproductive Health
To understand why spermidine matters so profoundly for fertility, we must first understand autophagy. The 2016 Medical Nobel Prize, awarded to Yoshinori Ohsumi for discoveries in autophagy, brought this cellular process into mainstream awareness. In reproductive medicine, its importance cannot be overstated.
The Basics of Autophagy
Autophagy is the cell’s quality control system — a highly conserved, tightly regulated process by which cells identify damaged proteins, dysfunctional organelles, lipid droplets, and other cellular debris; package them in double-membrane vesicles called autophagosomes; and deliver them to lysosomes for degradation and recycling of their components.
Think of autophagy as the cell’s combined recycling program and janitorial service. Without it, damaged components accumulate. This accumulation causes oxidative stress, disrupting cellular signaling, impairing energy metabolism, and ultimately triggering either cell dormancy or apoptosis (death). In cells that are meant to have extraordinary longevity and function like oocytes, autophagy is not optional. It is survival. Autophagy operates on multiple levels:
- Macroautophagy: The primary pathway — bulk or selective sequestration of cytoplasmic cargo into autophagosomes
- Mitophagy: Selective autophagy specifically targeting dysfunctional mitochondria — the pathway most relevant to fertility
- Chaperone-mediated autophagy (CMA): Direct translocation of individual proteins to the lysosome via chaperone proteins
- Selective autophagy of lipid droplets (lipophagy), ribosomes (ribophagy), peroxisomes (pexophagy), and pathogens (xenophagy)
Autophagy in Ovarian Biology
The ovary places extraordinary demands on cellular quality control. Each menstrual cycle, multiple follicles begin maturing under FSH stimulation, but typically only one reaches ovulation. The others undergo follicular atresia — a form of programmed, autophagy-regulated cell death in granulosa cells and oocytes that ensures only the most metabolically competent eggs advance. This selective pressure is nature’s own quality filter. Research consistently demonstrates that autophagy is essential for:
- Follicular development:
- The maturation of eggs from primordial follicles through primary, secondary, and antral stages to ovulatory readiness — a process taking months to years
- Oocyte quality:
- The cytoplasmic and nuclear maturity that determines whether a fertilized egg can successfully complete the first cell divisions and implant
- Granulosa cell survival:
- The supporting cells that nourish the oocyte depend on autophagy to survive the hypoxic conditions of follicular development
- Corpus luteum function:
- The post-ovulatory structure that produces progesterone essential for implantation requires autophagy for its development and eventual regression
- Ovarian aging resistance:
- Maintained autophagy slows the deterioration of ovarian reserve and oocyte quality with advancing age
As women age, ovarian autophagy becomes progressively impaired. Damaged mitochondria accumulate in oocytes, ATP production drops and reactive oxygen species (ROS) increase. The cellular environment becomes hostile to proper chromosome segregation during meiosis. The consequences are measurable and devastating. Aneuploidy rates in eggs rise from approximately 20% in women under 35 to over 80% in women over 42. These chromosomal errors represent failed implantations, miscarriages, and the heartbreaking reality of age-related infertility.
Autophagy in Spermatogenesis
Male fertility is equally dependent on robust autophagy, though the specific mechanisms differ. Sperm production involves dramatic, autophagy-dependent cellular remodeling. Starting from spermatogonial stem cells, germ cells must undergo meiosis to halve their chromosome number. Then spermiogenesis occurs — an extraordinary transformation in which round spermatids shed most of their cytoplasm, compact their DNA 1,000-fold using protamines, and build the flagellar machinery necessary for motility. Autophagy is essential at every stage:
- Spermatogonial stem cell (SSC) maintenance: Autophagy prevents SSC senescence and maintains the sperm-producing stem cell pool
- Meiotic progression: Autophagic clearance of damaged meiotic machinery enables proper chromosome segregation
- Spermiogenesis: Cytoplasmic removal during the spermatid-to-spermatozoon transition requires massive autophagic activity
- Residual body removal: Post-spermiation, Sertoli cells use autophagy to clear the cytoplasmic residual bodies shed by maturing sperm
- Sperm DNA quality: Autophagy prevents apoptosis in germ cells with damaged DNA, maintaining population quality
When autophagy is impaired by age, oxidative stress, toxin exposure, or declining spermidine levels, the entire spermatogenic process is compromised. Damaged mitochondria persist in sperm, ROS production increases, and sperm DNA fragmentation rises. This directly reduces fertilization capacity and embryo quality even when semen analysis parameters appear superficially normal.
Spermidine as a Master Autophagy Inducer
Spermidine induces autophagy through a well-characterized molecular mechanism: it inhibits the acetyltransferase EP300, which normally acetylates (and thereby inhibits) several pro-autophagy proteins including ATG7, LC3, and mTOR regulatory elements. By inhibiting EP300, spermidine allows these autophagy proteins to remain in their active, deacetylated state. They are essentially pressing the “on” switch for cellular recycling.
This mechanism is distinct from and complementary to other autophagy inducers (like rapamycin or caloric restriction). This makes spermidine particularly valuable. It activates autophagy through a pathway that doesn’t broadly suppress mTOR. This allows cells to continue growing and producing energy while simultaneously clearing their damaged components. For reproductive tissues under active development, this nuance matters considerably.
Key Autophagy Biomarkers to Know: LC3-II (lipidated form increases with autophagosome formation), Beclin-1 (initiates autophagy), p62/SQSTM1 (decreases when autophagy is active), ATG5/ATG7 (essential autophagy proteins). These markers appear throughout the research cited in this guide and are increasingly measured in fertility research to quantify cellular quality control activity.
Spermidine and Female Fertility: Oocyte Quality and Ovarian Aging
The evidence connecting spermidine to female fertility outcomes is among the most compelling in the preconception optimization literature. Multiple independent research groups, using diverse experimental models, have converged on the same conclusion: spermidine protects oocyte quality, reduces follicular atresia, and counteracts key mechanisms of ovarian aging — primarily through the activation of mitophagy.
Understanding Age-Related Oocyte Decline
To appreciate what spermidine can accomplish, we must first be precise about what goes wrong in aging oocytes. Advanced maternal age (typically defined as 35+ in fertility medicine) is the single most common reason women seek fertility support. But the problem isn’t simply that there are fewer eggs — it’s that the remaining eggs have accumulated decades of cellular damage. This compromises their ability to fertilize, develop, and implant successfully. The specific mechanisms of age-related oocyte decline include:
- Mitochondrial dysfunction:
- Oocytes in older women have fewer functional mitochondria with lower membrane potential, reduced electron transport chain efficiency, and lower ATP output — yet fertilization and the first cell divisions of the embryo require enormous energy expenditure
- Reduced mitophagy:
- Damaged mitochondria are not cleared efficiently, leading to accumulation of ROS-producing organelles within the oocyte cytoplasm
- Elevated oxidative stress:
- Excess ROS from dysfunctional mitochondria damages the meiotic spindle (causing chromosome segregation errors), cortical granules (affecting fertilization block mechanisms), and DNA
- Spindle assembly checkpoint (SAC) impairment:
- The cellular machinery that detects and corrects chromosome alignment errors during meiosis becomes less reliable with age
- Epigenetic dysregulation:
- DNA methylation and histone modification patterns that normally direct egg development become disordered
- Follicular atresia:
- Granulosa cells — which provide the oocyte with nutrients and signals — become more susceptible to oxidative stress-induced apoptosis, leading to premature follicle loss
These are not minor inefficiencies — they are fundamental cellular failures. Failures that produce the chromosomally abnormal eggs responsible for the vast majority of age-related IVF failures and pregnancy losses.
Landmark Research: Spermidine Rejuvenates Oocyte Quality via Mitophagy
Landmark Study: Zhang et al. (2023). ‘Spermidine restores maternal mitophagy and thereby improves oocyte quality and fertility in mice.’ Published in Nature Aging. Available at: https://pubmed.ncbi.nlm.nih.gov/
This landmark 2023 paper published in Nature Aging represents perhaps the most important single study connecting spermidine to female fertility. The research team used untargeted metabolomics — a comprehensive, unbiased analysis of all small molecules — on aged mouse ovaries. They identified spermidine as among the most critically depleted metabolites compared to young animals.
This was not a hypothesis-driven search for spermidine. The researchers cast the widest possible net and spermidine rose to the top. Having identified the deficit, they then experimentally confirmed causation by supplementing aged mice with spermidine and measuring the outcomes:
- Follicle development was significantly accelerated in supplemented animals
- Oocyte maturation rates improved substantially compared to unsupplemented aged controls
- Early embryonic development rates — measured through blastocyst formation — increased
- Overall female fertility (live birth rates in aged mice) increased with spermidine supplementation
Oocyte Quality
Crucially, the mechanism was precisely identified through microtranscriptomic analysis: spermidine activated mitophagy in aging oocytes, restoring mitochondrial function by clearing the damaged organelles that had accumulated over the animals’ lifespans. This wasn’t just correlation — it was a defined molecular pathway. Spermidine → mitophagy activation → damaged mitochondria cleared → ATP restored → oocyte quality improved.
The researchers also extended their findings to porcine oocytes exposed to oxidative stress. This model is considered far more translational to human physiology than mice alone. The protective effects were conserved, lending significant confidence to the human applicability of the findings.
For research-driven women navigating advanced maternal age, this study carries an important message: spermidine does not create new eggs. It cannot restore the follicles that have already been lost. What it can do is help the remaining oocytes perform better by restoring the mitochondrial housekeeping. It can reverse the mess that occurs with aging. This is precisely what advanced maternal age demands — not miracles, but optimized cellular function from the eggs that remain.
Additional Evidence: Follicular Atresia, Granulosa Cells, and Ovarian Protection
The Zhang et al. study is far from the only evidence. Multiple independent research groups have documented spermidine’s protective effects across multiple dimensions of ovarian biology:
5.3.1 Reduction of Follicular Atresia and Oxidative Damage
Reduction of Follicular Atresia and Oxidative Damage
A study available through PubMed Central (pmc.ncbi.nlm.nih.gov) documented that spermidine supplementation significantly reduced follicular atresia in an aged mouse model. The researchers measured comprehensive biomarkers of oxidative stress and autophagy in ovarian tissue:
- Antioxidant enzyme activity (SOD — superoxide dismutase, CAT — catalase, GSH-Px — glutathione peroxidase) was significantly higher in spermidine-supplemented animals
- Lipid peroxidation, measured by malondialdehyde (MDA) levels, was reduced — indicating less oxidative damage to cell membranes
- Autophagy proteins LC3 and Beclin-1 were upregulated, while p62 (which accumulates when autophagy is impaired) was reduced
- Polyamine balance in ovarian tissue was restored toward the profile of younger animals
The overall picture was of an ovarian microenvironment substantially more resistant to the oxidative damage that drives premature follicle loss — with more follicles surviving to contribute to the ovulatory pool.
Protection of Granulosa Cells
Granulosa cells — the support cells that surround and nourish each developing oocyte — are critical players in oocyte quality and are themselves highly susceptible to oxidative stress-induced apoptosis. Research using geese as a model for follicular dynamics demonstrated that spermidine alleviated oxidative stress-induced granulosa cell apoptosis specifically through the induction of autophagy, not simply through direct antioxidant action.
This distinction matters: spermidine doesn’t just neutralize the ROS already present. It restores the cellular machinery that prevents excessive ROS from accumulating in the first place.
Post-Ovulatory Oocyte Quality and IVF Relevance
Research on post-ovulatory aged porcine oocytes — directly relevant to IVF scenarios involving delayed egg retrieval or extended culture — demonstrated dramatic spermidine-dependent improvements (pubmed.ncbi.nlm.nih.gov). The findings included:
- Restoration of mitochondrial membrane potential (a direct measure of mitochondrial function)
- Significant reduction in intracellular ROS levels
- Inhibition of oocyte apoptosis (programmed cell death)
- Upregulation of autophagy markers including LC3 foci formation and lysosomal activity
- Improved spindle integrity — the chromosomal alignment structure critical for error-free cell division
- Better cortical granule distribution — affecting the fertilization block that prevents polyspermy
- Enhanced sperm-binding ability — improving fertilization rates
- Increased overall fertilization competence
These are the parameters fertility clinics measure to select the best embryos for transfer. Spermidine improved virtually all of them — in a model highly relevant to the human IVF setting.
Protection Against Environmental Toxin Exposure
Reproductive toxin exposure is an often-overlooked contributor to declining oocyte quality. Research on triclosan — an antimicrobial compound found in many personal care products and now recognized as an endocrine disruptor — demonstrated that spermidine protected porcine oocytes from triclosan-induced meiotic defects by attenuating oxidative stress-mediated apoptosis (researchsquare.com). Meiotic progression and fertilization competence were both preserved.
For women with occupational or lifestyle exposures to environmental chemicals — a group that includes many career-focused women in the 30–45 demographic — this protective capacity has direct practical relevance.
The Practical Takeaway for Women 35–45
Collectively, this body of research positions spermidine as a uniquely comprehensive protector of ovarian biology. It does not simply address one pathway — it acts simultaneously on mitochondrial quality control (mitophagy), oxidative stress (both direct antioxidant effects and indirect ROS reduction through mitophagy), granulosa cell survival, follicular atresia prevention, and oocyte maturation competence.
No other single dietary compound has demonstrated effects across this breadth of ovarian quality parameters. Combined with its excellent safety profile and the availability of highly bioavailable formulations, spermidine represents a genuinely novel and scientifically grounded approach to the challenge that defines reproductive medicine in the 21st century: aging egg quality.
Spermidine and Male Fertility: Addressing the Silent 55%
Male factor infertility contributes to approximately 55% of all infertility cases — yet men receive a disproportionately small fraction of the research attention, clinical resources, and supplement marketing directed at couples trying to conceive. A man is often offered a semen analysis, perhaps some vague lifestyle advice, and is otherwise expected to be a supportive bystander to his partner’s medical journey. This is scientifically inadequate and increasingly unacceptable to the generation of men now entering their prime reproductive years.
The good news is that male fertility — in most cases — is highly responsive to intervention. Unlike women, who must work with the oocytes they were born with, men produce entirely new sperm every 74 days. This means that a man who optimizes his cellular environment today will be producing measurably improved sperm within a single spermatogenic cycle. Spermidine is one of the most scientifically grounded interventions for that optimization.
The Spermatogenesis Process and Its Vulnerabilities
Understanding why spermidine helps requires understanding what sperm production actually involves. Spermatogenesis is one of the most metabolically demanding processes in the body — an extraordinary cellular transformation that takes approximately 74 days in the seminiferous tubules of the testes, followed by an additional 12–21 days of epididymal maturation. The process unfolds in three stages:
- Proliferative phase: Spermatogonial stem cells (SSCs) divide mitotically, maintaining the stem cell pool while generating committed progenitor cells
- Meiotic phase: Primary spermatocytes undergo two meiotic divisions to produce haploid round spermatids with 23 chromosomes
- Spermiogenic phase: Round spermatids undergo radical morphological transformation — nuclear compaction, acrosome development, flagellum construction, cytoplasm shedding — to become mature spermatozoa
Each stage is autophagy-dependent, and each stage is vulnerable to the same enemies: oxidative stress, mitochondrial dysfunction, DNA damage, and the progressive decline in spermidine that comes with age.
Mature sperm are particularly vulnerable to oxidative stress for several reasons unique to their biology: they contain extremely high concentrations of polyunsaturated fatty acids (PUFAs) in their membranes — particularly DHA — which are highly susceptible to lipid peroxidation; they have minimal cytoplasm and thus minimal antioxidant enzyme defense; and once mature, they have essentially no capacity for DNA self-repair. This means that oxidative damage sustained in the epididymis — where sperm mature and are stored — cannot be corrected.
Spermidine’s Protective Mechanisms in Male Fertility
Spermidine targets the biological vulnerabilities of sperm production through several distinct but complementary mechanisms:
Oxidative Stress Reduction
ROS from lifestyle exposures (smoking, alcohol, environmental toxins), elevated scrotal temperature, sedentary habits, and age-related mitochondrial inefficiency are the primary drivers of the sperm DNA fragmentation index (DFI) — the clinically measured marker of sperm DNA integrity. Elevated DFI (typically defined as >15–25% fragmentation, depending on the test) correlates with reduced fertilization rates, impaired embryo development, and increased miscarriage risk, even when basic semen parameters appear normal.
Spermidine reduces sperm oxidative stress through its dual action as a direct antioxidant (scavenging certain reactive species) and as a mitophagy inducer that removes the damaged mitochondria generating excess ROS in the first place. Research specifically documenting spermidine’s ability to reduce sperm oxidative stress has been published in specialized nutraceutical and reproductive science literature, correlating reduced ROS with improved sperm parameters across multiple measures.
Mitochondrial Function and ATP for Motility
Sperm motility — the ability to swim with the forward, progressive movement necessary to navigate the female reproductive tract and penetrate the egg — is entirely dependent on ATP produced by the mitochondria clustered in the sperm midpiece. These midpiece mitochondria power the dynein ATPases that drive the flagellar beat.
When mitochondrial function is impaired — whether by accumulated oxidative damage, reduced mitophagy, or declining spermidine — ATP output drops, flagellar beat frequency decreases, and progressive motility falls. Spermidine’s enhancement of mitophagy and mitochondrial biogenesis directly addresses this mechanism, supporting the energy infrastructure that sperm motility requires.
Sperm Morphology and Structural Integrity
Sperm morphology — the percentage of sperm with normal head, midpiece, and tail structure — reflects the quality of the spermiogenic transformation from round spermatid to mature spermatozoon. Proper morphology requires precisely regulated autophagy to shed cytoplasmic droplets, form the acrosomal cap, and align the nuclear-cytoskeletal complex correctly.
Higher spermidine levels in testicular tissue correlate with higher percentages of morphologically normal sperm forms. By supporting the autophagic spermiogenic remodeling process, spermidine helps ensure that the resulting mature sperm have the structure necessary to reach and fertilize the egg.
Protection Against Toxin-Induced Testicular Dysfunction
A compelling demonstration of spermidine’s testicular protective capacity comes from research on triptolide-induced spermatogenesis impairment (pubmed.ncbi.nlm.nih.gov). Triptolide is a plant compound known to severely damage testicular tissue and impair spermatogenesis. In this experimental model, restoring polyamine levels — including spermidine — reversed sperm count drops, histological testicular damage, reproductive hormone imbalances, and oxidative stress. The protective mechanism involved HSP70 upregulation (a cellular stress-protective protein) and gut microbiota support.
While triptolide exposure is not a common human scenario, the model demonstrates that spermidine can rescue spermatogenesis from severe chemically-induced impairment — suggesting broadly protective capacity against the more common and diffuse environmental exposures (BPA, phthalates, pesticides) that accumulate in the real world.
The 74-Day Strategic Window
Understanding the spermatogenesis timeline transforms how men should approach preconception supplementation. The implication of the 74-day cycle is that:
- Any intervention — positive or negative — takes approximately 2.5 months to manifest in ejaculated sperm
- A man who begins spermidine supplementation today will produce genuinely improved sperm in about 10–12 weeks
- Conversely, a man who smokes, drinks heavily, or experiences heat exposure to the testes today will see the consequences in his semen analysis 2.5 months later
- Pre-IVF sperm optimization should begin at minimum 3 months before sperm collection, with 4–6 months preferred
This defined, predictable timeline is actually empowering — it transforms male fertility from a fixed biological state into a dynamic parameter that responds to deliberate optimization. And for career-focused, health-conscious men in their 30s and 40s who respond to evidence and timelines, this framing resonates. The 74-day window is not a limitation — it’s a roadmap.
Oxidative Stress: The Common Enemy of Egg and Sperm Quality
No discussion of reproductive cellular biology is complete without a thorough examination of oxidative stress — the single most significant modifiable factor affecting both egg and sperm quality, and the primary target of spermidine’s fertility-protective actions.
What Is Oxidative Stress and Why Do Reproductive Cells Suffer?
Oxidative stress is the imbalance between the production of reactive oxygen species (ROS) — chemically reactive molecules containing oxygen, including superoxide radical, hydrogen peroxide, and hydroxyl radical — and the body’s antioxidant defenses. ROS are normal byproducts of cellular metabolism, particularly mitochondrial respiration. The problem arises when their production exceeds the capacity of antioxidant systems (superoxide dismutase, catalase, glutathione peroxidase, vitamin E, vitamin C, etc.) to neutralize them. Reproductive cells are peculiarly susceptible to oxidative stress damage for reasons rooted in their unique biology:
- Oocytes have some of the highest mitochondrial densities in the human body — necessary to power fertilization and early embryonic development, but also generating significant ROS as a metabolic byproduct
- Sperm membranes are exceptionally rich in polyunsaturated fatty acids — essential for membrane fluidity and fertilization competence but highly vulnerable to lipid peroxidation by ROS
- The blood-testes barrier creates a specialized but also relatively vulnerable microenvironment in the testes
- Granulosa cells surrounding each developing follicle must survive in conditions of changing oxygenation and hormonal flux — creating ongoing oxidative challenge
The consequences of unchecked oxidative stress in reproductive cells are well-documented and devastating: chromosomal fragmentation in oocytes (leading to aneuploidy), DNA strand breaks in sperm, mitochondrial membrane damage, acrosomal enzyme inactivation, and membrane lipid peroxidation that impairs capacitation and the acrosome reaction. Up to 80% of men with infertility have elevated levels of seminal ROS — a staggering figure that underscores oxidative stress as a primary driver of male factor infertility.
How Spermidine Addresses Oxidative Stress at Multiple Levels
Spermidine is unusual among fertility-relevant compounds in that it addresses oxidative stress through multiple distinct mechanisms, rather than simply providing antioxidant electrons:
- Direct radical scavenging: Spermidine’s amino groups can directly neutralize certain reactive oxygen and nitrogen species, providing immediate antioxidant protection
- Mitophagy-mediated ROS source reduction: By activating mitophagy and clearing dysfunctional mitochondria — the primary intracellular source of excess ROS — spermidine addresses the problem at its origin rather than just neutralizing downstream products
- Mitochondrial biogenesis enhancement: Spermidine promotes the generation of new, healthy mitochondria to replace those cleared by mitophagy, restoring cellular energy capacity while reducing ROS output
- Antioxidant enzyme upregulation: Spermidine supplementation has been shown to increase the activity of SOD, CAT, and GSH-Px — the cellular antioxidant enzymes — in ovarian and testicular tissue
- Anti-inflammatory effects: Chronic low-grade inflammation generates ROS and degrades reproductive tissue microenvironments; spermidine modulates NF-κB and other inflammatory signaling pathways to reduce this sustained oxidative burden
This multi-level attack on oxidative stress — simultaneously reducing its production, enhancing its neutralization, and improving the cellular infrastructure for future resilience — is what distinguishes spermidine from simpler antioxidant supplements like Vitamin C or E, which primarily address only the third mechanism above.
Oxidative Stress, Aging, and the Fertility Cliff
Age amplifies every dimension of oxidative stress in reproductive cells. Mitochondrial efficiency decreases with age (the “aging mitochondria” phenomenon), meaning more ROS is produced per unit of ATP generated. At the same time, antioxidant enzyme activity — SOD, CAT, GSH-Px — declines with age. And the accumulation of oxidative damage from decades of ROS exposure means older reproductive cells are starting from a compromised baseline.
This is why the decline in fertility accelerates after 35 (in women) and the 30s–40s (in men) rather than declining linearly from the reproductive peak. Oxidative stress compounds, mitophagy declines, spermidine levels drop, and the cellular environment of the gonads deteriorates in a self-reinforcing cascade. Restoring spermidine is not merely addressing one link in this chain — it interrupts the cascade at a central hub, simultaneously improving multiple downstream outcomes.
Bioavailability and Liposomal Spermidine: Why Delivery Method Is Critical
Understanding the science of spermidine’s fertility benefits is only half the equation. The other half — the practical half that determines whether supplementation actually works — is bioavailability: the percentage of an ingested compound that reaches its target tissues in an active form. For spermidine, this is where formulation science becomes essential.
The Problem with Standard Oral Spermidine
Spermidine taken in standard oral form faces significant pharmacokinetic obstacles:
- First-pass gut metabolism: Spermidine is actively metabolized by intestinal bacteria and gut epithelial enzymes before reaching systemic circulation. SSAT (spermidine/spermine N1-acetyltransferase) in the gut wall rapidly converts spermidine to less active metabolites
- Rapid systemic catabolism: Once absorbed, spermidine is quickly converted to spermine or catabolized back to putrescine by circulating enzymes, limiting the time window for tissue uptake
- Limited tissue penetration: Even absorbed spermidine may not achieve sufficient concentrations in highly vascular-selective tissues like ovarian follicles or the blood-testes barrier-protected seminiferous tubules
- Dose-response complexity: Higher oral doses trigger increased SSAT activity, paradoxically accelerating catabolism and limiting the benefit of dose increases
The clinical implication is that blood and salivary spermidine levels often do not rise significantly even at quite high standard oral doses. This is not a trivial problem — it means that much of the research benefit observed in animal studies (where spermidine was often administered in ways that bypass first-pass metabolism) may not be replicated with standard oral supplementation in humans.
Liposomal Delivery: The Solution
Liposomal drug delivery technology addresses the bioavailability challenge through an elegant biological solution. Spermidine molecules are encapsulated within phospholipid bilayer vesicles — liposomes — that are structurally identical to cell membranes. This encapsulation provides multiple advantages:
- Protection from premature degradation: The phospholipid shell protects spermidine from gut enzymes and bacterial metabolism during transit through the GI tract
- Enhanced intestinal uptake: Liposomes fuse with intestinal epithelial membranes, delivering their contents directly into enterocytes, bypassing the luminal degradation that limits standard spermidine absorption
- Improved lymphatic absorption: Larger liposomal particles enter the lymphatic system rather than portal circulation, partially bypassing first-pass hepatic metabolism
- Direct cellular delivery: Once in systemic circulation, liposomes can fuse with target cell membranes, delivering spermidine intracellularly where it needs to be to activate autophagy
- Controlled release: Liposomal formulations can be designed to release their payload over time, maintaining more consistent tissue spermidine levels
Advanced liposomal spermidine formulations have reported substantially improved bioavailability compared to standard oral preparations — with some formulations claiming cellular uptake improvements of several-fold over conventional supplements. For fertility applications, where the target tissues — ovarian granulosa cells, oocytes, testicular Sertoli and germ cells — are protected by biological barriers, this enhanced tissue penetration is not merely a commercial advantage but a functional prerequisite for clinical effectiveness.
What to Look for in a Spermidine Supplement
For couples investing in preconception supplementation, formulation quality is as important as the underlying science. Key considerations:
- Delivery system: Liposomal formulations are preferred over standard oral supplements. Look for phosphatidylcholine-based liposomes with particle sizes <200 nm for optimal absorption
- Spermidine concentration: Research doses have ranged from 1–10 mg/day. For fertility optimization, 3–10 mg/day of bioavailable spermidine is a reasonable target based on current evidence
- Source purity: Wheat germ extract is the most common natural spermidine source. Quality products will specify the extraction method and provide third-party purity testing
- Bioavailability documentation: Look for brands that have conducted or cite pharmacokinetic studies demonstrating actual blood/tissue level increases, not just in vitro cell studies
- Third-party testing: Independent verification of label claims for heavy metals, pesticides, and actual spermidine content
Editorial Note: The technology behind liposomal delivery is well-established, but quality varies significantly among manufacturers. Couples should research specific products using the criteria above, and always disclose supplement use to their healthcare providers.

The Autophagy-Optimized Conception Protocol (AOCP)
The following evidence-informed protocol synthesizes the research on spermidine, autophagy, oxidative stress, and preconception optimization into a practical 3–6 month framework for couples preparing to conceive — whether naturally or through assisted reproduction. This is not a medical protocol; it is a scientifically grounded lifestyle and supplementation framework that should be discussed with your reproductive endocrinologist or OB-GYN before implementation.
Timeline Overview: This protocol requires a minimum of 3 months to capture the full 74-day spermatogenic cycle (for men) plus a buffer, and to allow the cumulative autophagy-inducing effects to improve ovarian follicular quality (for women, optimal benefit at 4–6 months). Start as early as possible before your target conception date.
Phase 1: Baseline Assessment (Weeks 1–2)
Establish objective starting points before beginning supplementation. This enables genuine outcome tracking and informs individualized protocol adjustments.
For Men:
- Complete semen analysis (count, motility, morphology, volume, pH)
- Sperm DNA fragmentation index (DFI) — highly recommended, underutilized
- Reproductive hormones: FSH, LH, testosterone, estradiol, prolactin
- Oxidative stress markers if available (seminal ROS, 8-OHdG)
- Metabolic baseline: fasting glucose, insulin, HbA1c (metabolic dysfunction impairs spermatogenesis)
For Women:
- Ovarian reserve markers: AMH (anti-Müllerian hormone), FSH (day 3), antral follicle count (AFC) via ultrasound
- Hormonal assessment: estradiol, LH, progesterone (timed to cycle)
- Thyroid function: TSH, free T4 (thyroid dysfunction profoundly affects fertility)
- Inflammatory markers: CRP, if available
- Vitamin D level (significant proportion of fertility patients are deficient)
Phase 2: Daily Foundation (Months 1–4 Minimum)
The core protocol runs continuously through conception. Consistency is essential — the research benefits of spermidine accrue over weeks and months, not days.
Liposomal Spermidine Supplementation
Liposomal spermidine is the cornerstone of the AOCP. Dosing: 3–10 mg/day of bioavailable spermidine (follow specific product guidance, as total product dose varies by formulation). Take consistently — split dosing (morning and evening) may maintain more stable tissue levels. Take with or without food based on product instructions (liposomal formulations are generally more food-independent than standard forms).
Spermidine-Rich Dietary Foundation
Dietary spermidine is synergistic with supplementation — it contributes additional substrate to the polyamine pool and promotes gut microbiome health that supports endogenous polyamine production. Daily targets:
- 2–3 tablespoons wheat germ (can add to yogurt, smoothies, oatmeal, or baked goods)
- 1 serving soybeans, edamame, or tempeh
- 1–2 servings mushrooms (shiitake, oyster, or cremini are highest in spermidine)
- 1 serving legumes (chickpeas, lentils, black beans)
- Aged cheese in moderation (if dairy is tolerated)
Evidence-Based Synergistic Supplements
These compounds work through mechanisms complementary to spermidine, addressing the same cellular vulnerabilities through different but synergistic pathways:
| Supplement | Dose and Rationale |
| CoQ10 (Ubiquinol form) | 200–600 mg/day — supports mitochondrial electron transport; extensive fertility research, particularly for oocytes |
| Omega-3 fatty acids (DHA/EPA) | 2–3 g/day — reduces inflammation, supports sperm membrane integrity, improves egg quality |
| NAC (N-Acetyl Cysteine) | 600 mg/day — precursor to glutathione, the master cellular antioxidant; reduces oxidative stress |
| Vitamin D (D3) | Optimize to 50–70 ng/mL — deficiency associated with reduced IVF success; aim for sufficiency, not megadosing |
| Vitamin E (mixed tocopherols) | 200–400 IU/day — lipophilic antioxidant; protects PUFAs in sperm membranes and oocytes |
| Folate / Methylfolate | 400–800 mcg/day — essential for DNA synthesis; critical in preconception and early pregnancy |
| Zinc (for men) | 25–30 mg/day — essential for testosterone production, sperm maturation, and antioxidant enzyme activity |
| Myo-Inositol (for women with PCOS) | 2–4 g/day — improves insulin sensitivity and oocyte quality, particularly in PCOS-associated infertility |
Lifestyle Pillars
Supplements cannot compensate for foundational lifestyle factors. These behaviors directly modulate autophagy, oxidative stress, and reproductive tissue cellular health:
- Exercise: 150 minutes per week of moderate-intensity activity, incorporating both aerobic training (cardiovascular and mitochondrial health) and resistance training (testosterone optimization for men, metabolic health for both). Avoid extremes — over-training elevates cortisol and suppresses reproductive hormones.
- Sleep: 7–9 hours per night. Growth hormone — secreted primarily during deep sleep — is a potent autophagy activator. Chronic sleep deprivation directly impairs spermatogenesis and disrupts menstrual regularity.
- Stress management: Chronic cortisol elevation suppresses autophagy, impairs LH pulsatility (reducing testosterone in men and disrupting ovulation in women), and generates oxidative stress. Evidence-based practices: mindfulness meditation, yoga, HRV biofeedback, therapeutic support.
- Avoid testicular heat (men): Spermatogenesis requires a temperature 2–4°C below core body temperature. Avoid hot tubs, saunas, laptops on lap, tight underwear, and prolonged sitting for extended periods during the optimization window.
- Minimize alcohol and eliminate smoking: Both are well-documented gonadotoxins. Alcohol disrupts testosterone production, impairs antioxidant status, and interferes with estrogen metabolism. Smoking is one of the most potent accelerators of ovarian aging identified.
- Reduce plastic and endocrine disruptor exposure: BPA, phthalates, parabens, and certain pesticides disrupt reproductive hormones and generate oxidative stress. Choose glass or stainless steel containers, organic produce where practical, and fragrance-free personal care products.
Phase 3: Autophagy Amplification (Strategic Windows)
For couples who want to augment spermidine’s autophagy-inducing effects, periodic implementation of time-restricted eating provides additional benefit without compromising nutritional status during active preconception preparation:
- 12–14 hour overnight fasting (e.g., last meal at 7 PM, first meal at 7–9 AM): This gentle restriction activates autophagy and improves insulin sensitivity without significant caloric restriction. Suitable for most women throughout the preconception period.
- 16:8 intermittent fasting (16 hours fasting, 8-hour eating window): More potent autophagy induction; appropriate for men throughout the protocol and for women in the earlier months of preconception preparation, before active conception attempts begin.
- Periodic Fasting-Mimicking Diet (FMD) cycles: A specific 5-day protocol of very low-calorie, specific macronutrient composition that has been shown in human trials to significantly improve autophagic flux markers, reduce IGF-1, improve insulin sensitivity, and elevate ketone bodies (See Section 11 for full discussion). Consider 1–2 cycles during the preconception preparation phase, but not during active conception attempts.
Phase 4: Monitoring and Optimization (Month 3 and Beyond)
Track progress and adjust the protocol based on objective data and subjective response:
- Men: Repeat semen analysis and DFI at 3 months. Look for improvements in progressive motility (>40% threshold), morphology (Kruger strict criteria), total motile sperm count, and DFI reduction.
- Women: Discuss repeat AMH and AFC with your doctor at 3–6 months. Track menstrual cycle regularity, flow quality, and any changes in PMS or cycle symptoms as indirect quality-of-life indicators.
- Both: Track energy levels, sleep quality, and general wellbeing — spermidine’s autophagy-inducing effects benefit mitochondrial health broadly, and many people report improved energy and recovery as collateral benefits.
- Maintain clinical transparency: All supplements should be disclosed to your fertility specialist, particularly if beginning IVF stimulation. Spermidine’s mechanism of action is complementary to IVF protocols, but your specialist should be informed.
Cost Reality Check
A full IVF cycle with medications typically costs $18,000–$30,000 and may require multiple attempts. The AOCP, properly implemented with high-quality liposomal spermidine and the synergistic supplements above, costs a small fraction of that — and potentially reduces the number of IVF cycles required by improving the cellular quality of the eggs and sperm used in treatment. At the very least, it optimizes the biological environment for natural conception.
70% of people undergoing fertility treatments already use supplements, yet 45% do not disclose this to their physicians. Please be transparent with your fertility team. Spermidine is safe and complementary to standard care — but your doctors need to know what you are taking.

Spermidine and IVF: The 1% Insurance Policy
For the substantial portion of couples in the fertility optimization community who are pursuing or preparing for IVF, spermidine’s role deserves specific attention. IVF is extraordinary technology — it can achieve pregnancies that would otherwise be biologically impossible. But it also has profound limitations that science and technology cannot currently overcome.
What IVF Cannot Fix
IVF clinics have become extraordinarily sophisticated in their ability to stimulate follicle development, retrieve eggs, fertilize them in controlled laboratory conditions, assess embryo morphology and increasingly chromosomal status through PGT-A (preimplantation genetic testing for aneuploidies), and time embryo transfer for maximum implantation potential.
What they cannot do is repair fundamental cellular quality deficits before retrieval. A laboratory cannot restore mitochondrial membrane potential in an egg that has accumulated decades of mitophagy-impaired oxidative damage. It cannot repair fragmented DNA in sperm. It cannot replace the cytoplasmic competence that determines whether a fertilized embryo can successfully complete the first cell divisions. These biological fundamentals must be addressed before IVF — through preconception optimization.
The most common cause of IVF failure in women over 35 is chromosomal abnormality in retrieved eggs — not laboratory quality, not the skill of the embryologist, not the timing of transfer. The eggs themselves are compromised. This is why improving the cellular environment of oocyte development in the months before stimulation is the highest-leverage intervention available to women preparing for IVF.
How Spermidine Complements IVF
The research directly relevant to IVF outcomes includes:
- Post-ovulatory oocyte quality studies demonstrating spermidine’s preservation of spindle integrity, cortical granule distribution, and fertilization competence — directly predictive of IVF embryo development outcomes
- Documentation of reduced aneuploidy rates correlating with improved oocyte mitochondrial function — the mechanism spermidine directly targets
- Sperm parameter improvements that predict better fertilization rates in IVF and ICSI procedures
- Protection against environmental toxin-induced oocyte damage — relevant for women with occupational exposures or elevated body burden of endocrine disruptors
- Granulosa cell survival improvements that may translate to better follicular development during stimulation
Spermidine’s mechanism — autophagy induction — is unlikely to interfere with standard IVF pharmacological protocols, which use gonadotropins to override normal follicular selection and stimulate multiple follicle development simultaneously. Autophagy enhancement works at the subcellular level within eggs and sperm, not at the level of hormonal signaling that IVF drugs target.
However, as with any supplement during an IVF cycle, full disclosure to your reproductive endocrinologist is mandatory. Some clinics have begun to acknowledge autophagy enhancers as rational adjuncts; others prefer a more conservative approach. Your specialist’s guidance, informed by knowledge of your specific protocol and health history, takes precedence.
The Economics of Optimization
Consider this calculation: A 3-month supply of high-quality liposomal spermidine, combined with CoQ10 and the other synergistic supplements in the AOCP, costs approximately $200–$400 — roughly 1–2% of a single IVF cycle’s cost. If this investment improves egg quality sufficiently to increase the probability of retrieving more usable blastocysts, reduce the number of cycles required, or improve implantation rates — the return on investment is extraordinary, measured in both financial terms and the incalculable emotional cost of failed cycles.
Even if the impact is more modest — incrementally improving the cellular environment without producing dramatic measurable changes in specific parameters — the risk-benefit calculation remains strongly favorable given spermidine’s excellent safety profile and the low cost relative to the investment in IVF itself.
The Fasting-Mimicking Diet: Amplifying Autophagy Naturally
Caloric restriction and fasting are among the most powerful natural autophagy inducers identified, operating through mechanisms that are distinct from and complementary to spermidine. When nutrient availability is reduced, mTOR (mechanistic target of rapamycin) — the master suppressor of autophagy — becomes inhibited, releasing the brake on cellular recycling. AMPK (AMP-activated protein kinase), a cellular energy sensor, simultaneously activates autophagy-promoting pathways.
The Fasting-Mimicking Diet (FMD) Evidence
A pivotal human pilot trial published on PubMed (pubmed.ncbi.nlm.nih.gov) demonstrated that a structured Fasting-Mimicking Diet protocol significantly improved multiple markers of biological aging and cellular health:
- Significantly improved autophagic flux markers — measurable activation of cellular recycling
- Reduced IGF-1 (insulin-like growth factor 1) — elevated IGF-1 suppresses autophagy and is associated with reduced longevity and increased cancer risk
- Improved insulin sensitivity — impaired insulin sensitivity is a direct contributor to PCOS and ovarian aging
- Elevated circulating ketone bodies — ketones serve as alternate fuel sources that support mitochondrial health and may themselves induce autophagy
The FMD protocol, developed by Dr. Valter Longo at the Longevity Institute at USC, typically involves 5 days of very low calorie intake (700–800 calories/day on day 1, 500 calories/day on days 2–5) with a specific macronutrient composition (low protein, moderate healthy fats, limited carbohydrates) that mimics the metabolic state of fasting while still providing some nutrition.
FMD and Fertility: A Nuanced Picture
For couples actively trying to conceive, the relationship between fasting and fertility requires careful navigation. The female reproductive system is exquisitely sensitive to energy availability. The brain’s GnRH (gonadotropin-releasing hormone) pulse generator — which drives the entire hormonal cascade of the menstrual cycle — is one of the first systems to shut down under conditions of energy deficit. Excessive caloric restriction can suppress ovulation entirely (hypothalamic amenorrhea) and impair embryo implantation.
This creates an important practical distinction: aggressive fasting protocols are inappropriate for women in the conception window (i.e., around ovulation or during the luteal phase) or for women with very low BMI, history of disordered eating, or clinical hypothalamic amenorrhea.
However, strategic use of milder fasting approaches during the preconception optimization phase — before active conception attempts begin — is both reasonable and potentially valuable:
- 1–2 FMD cycles during the preconception preparation phase (3–6 months before active trying) provide significant autophagy activation and metabolic recalibration
- 12–14 hour overnight fasting (eating between 8 AM and 8 PM, for example) is gentle enough to be sustained continuously without reproductive impact, while still providing meaningful autophagy support
- For men, more aggressive time-restricted eating (16:8) can be maintained throughout the preconception optimization phase without reproductive concern
Crucially, spermidine supplementation provides autophagy benefits without requiring caloric restriction — making it particularly valuable for women who cannot or should not fast aggressively. Spermidine acts through the EP300 inhibition pathway rather than the mTOR/AMPK pathway, meaning its autophagy-inducing effects are additive to fasting-induced autophagy and operate independently of caloric status.
Safety, Dosing, and Practical Implementation
Any fertility supplement recommendation must address safety with full transparency. On this dimension, spermidine has an unusually reassuring profile that distinguishes it from many of the more aggressive interventions being explored in fertility medicine.
Safety Profile
- Endogenous compound: Spermidine is naturally produced in every human cell and is present in many common foods that have been consumed safely for millennia. It is not a novel pharmaceutical compound introducing foreign chemistry — it is restoring a molecule the body already uses.
- Long-term human safety data: Multiple longitudinal studies in humans have assessed spermidine supplementation for longevity and cognitive aging applications without identifying serious adverse effects at supplemental doses.
- Broad mechanism: Autophagy induction is a fundamental cellular housekeeping process. Enhancing a process the body uses continuously for cellular maintenance is inherently safer than blocking a pathway (as with rapamycin) or introducing a foreign signaling molecule.
- GI tolerance: The most commonly reported side effect with oral spermidine supplementation is mild gastrointestinal adjustment at higher doses. This typically resolves within a few days and is minimized by starting with a lower dose and titrating up.
- No documented negative interactions: No known adverse interactions with prenatal vitamins, standard fertility medications, or commonly used supplements have been identified. However, this absence of documented interactions is not a substitute for disclosure to your healthcare provider.
Dosing Considerations for Fertility Applications
Research studies have used a range of dosing protocols. For fertility optimization specifically, current evidence suggests:
| Parameter | Guidance |
| Standard supplemental dose | 1–3 mg/day (basic cellular support) |
| Fertility optimization dose | 3–10 mg/day bioavailable spermidine |
| Research study range | 1–10 mg/day in human studies |
| Dietary contribution | ~2–5 mg/day with optimized diet |
| Duration before expecting gamete improvement | 3 months minimum (sperm); 4–6 months (oocytes) |
| Formulation preference | Liposomal strongly preferred for fertility applications |
Note that “dose” in liposomal products refers to bioavailable spermidine, which is inherently lower per capsule than the total content in standard supplements. A product containing 10 mg of liposomal spermidine may deliver more active compound to reproductive tissues than a product containing 50 mg of standard spermidine.
Special Considerations
- IVF patients: Disclose all supplements to your reproductive endocrinologist before beginning stimulation. Discuss timing of any planned cessation around the egg retrieval period if your clinic prefers a supplement-free window.
- Individuals with metabolic disorders: Those with disruptions in polyamine metabolism (rare but existing conditions) should consult with a metabolic specialist before spermidine supplementation.
- Interactions with specific medications: Spermidine is generally safe alongside most medications, but individuals on chemotherapy agents (which may interact with polyamine biosynthetic pathways) should consult their oncologist.
- Pregnancy: Evidence for spermidine supplementation during active pregnancy is limited. Many practitioners recommend continuing through early trying-to-conceive but transitioning guidance to a prenatal-focused protocol once pregnancy is confirmed. Discuss with your OB-GYN.
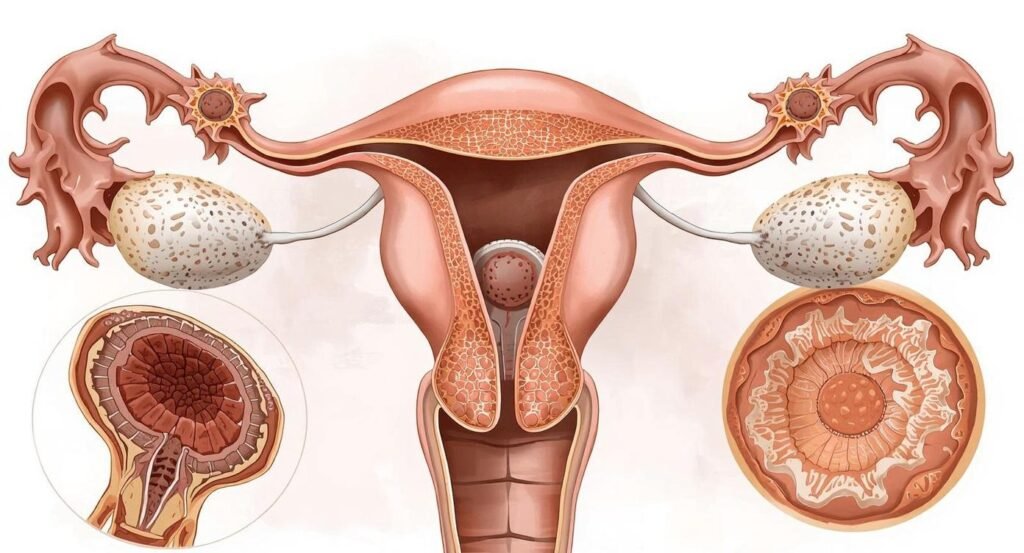
The Future of Fertility Science: What’s Coming Next
Spermidine’s emergence as a fertility compound reflects a broader paradigm shift in reproductive medicine — from organ-level interventions (hormonal stimulation, surgical correction) to cellular-level optimization. Several parallel developments suggest this trend will accelerate dramatically in the coming decade:
Converging Technologies in Preconception Optimization
- NAD+ precursors (NMN and NR): Nicotinamide mononucleotide and nicotinamide riboside restore declining NAD+ levels, supporting mitochondrial function and SIRT1-mediated autophagy activation. Research on NMN for oocyte quality has shown promise, and its synergy with spermidine — each activating autophagy through distinct mechanisms — makes their combination logically attractive.
- Senolytics for gonadal rejuvenation: Senescent cells accumulate in the ovaries and testes with age, secreting inflammatory SASP (senescence-associated secretory phenotype) factors that damage the reproductive microenvironment. Compounds that selectively clear senescent cells (dasatinib, quercetin, fisetin) are being actively researched for ovarian aging applications.
- Mitochondrial replacement therapy: Experimental techniques including spindle transfer and pronuclear transfer — which replace dysfunctional mitochondria in aging eggs with those from young donor eggs — highlight the centrality of mitochondrial health to oocyte quality. Spermidine’s natural mitophagy approach addresses the same problem non-invasively.
- Personalized fertility genomics: Genetic variants in autophagy genes, antioxidant enzymes, and polyamine biosynthetic pathways are increasingly recognized as modifying individual fertility outcomes. Personalized preconception protocols based on genetic profile are becoming feasible.
- Spermidine-specific fertility RCTs: The mechanistic and animal data supporting spermidine for fertility are robust; what the field currently lacks are large, well-designed randomized controlled trials measuring live birth rates in humans. Multiple such trials are in design or early stages. Their results — expected in the next 3–7 years — will likely cement or refine the evidence base presented in this guide.
Fertility Science
Spermidine is not the final word in fertility optimization science — it is an early, compelling entry in a field that is developing rapidly. As personalized genomics, expanded biomarker panels, and increasingly refined preconception protocols emerge, the underlying principles this guide presents — autophagy, mitochondrial health, oxidative stress management — will remain central. The tools will improve; the biological targets will not change.
Frequently Asked Questions (FAQ)
The following questions represent the most common queries from couples researching spermidine for fertility. This section is structured for FAQ schema markup for SEO purposes.
Can I take spermidine while on IVF medications?
Can I take spermidine while on IVF medications?
Yes — spermidine is generally compatible with standard IVF medications, as it operates at the subcellular autophagy level rather than through the hormonal signaling pathways that gonadotropins target. However, full disclosure to your reproductive endocrinologist is mandatory before beginning any supplement regimen during an IVF cycle. Some clinics prefer a supplement-free period around egg retrieval; follow your specialist’s guidance.
How long does it take to see results from spermidine supplementation?
How long does it take to see results from spermidine supplementation?
For sperm quality improvements, expect measurable changes in 10–12 weeks (one full spermatogenic cycle), with optimal improvements appearing at 3–4 months. For female fertility benefits, the timeline is longer because follicular development is slower — meaningful improvements in oocyte quality require 4–6 months of consistent supplementation, though ovarian tissue health may begin improving sooner. Consistency is essential; the benefits of spermidine are cumulative.
Is dietary spermidine enough, or do I need to supplement?
Is dietary spermidine enough, or do I need to supplement?
Optimizing dietary spermidine intake — through wheat germ, soybeans, mushrooms, legumes, and aged cheeses — is genuinely helpful and supports the supplementation protocol. However, achieving the consistent elevated systemic levels shown in research to meaningfully activate autophagy in reproductive tissues is unlikely with diet alone, particularly given age-related increases in spermidine catabolism. High-quality liposomal supplementation is recommended for fertility optimization applications.
Does spermidine help with male fertility, or is it mainly for women?
Does spermidine help with male fertility, or is it mainly for women?
Spermidine has robust evidence supporting benefits for both male and female fertility through distinct but complementary mechanisms. For men, it addresses the primary driver of male factor infertility — oxidative stress — while also supporting mitochondrial function for motility and protecting sperm DNA integrity. Male fertility is responsive to intervention on a defined 74-day timeline, making spermidine particularly actionable for men in the preconception phase.
Are there any interactions between spermidine and prenatal vitamins?
Are there any interactions between spermidine and prenatal vitamins?
No known negative interactions exist between spermidine and standard prenatal vitamins. They are complementary: spermidine supports preconception cellular optimization while prenatal vitamins address the micronutrient requirements of early pregnancy. Many practitioners recommend beginning prenatals 3 months before conception and continuing throughout pregnancy; spermidine can run concurrently during the preconception phase.
Can spermidine improve egg quality after 40?
Can spermidine improve egg quality after 40?
The research in aged animal models demonstrates meaningful improvements in oocyte mitochondrial function, ROS levels, fertilization rates, and embryonic development through spermidine supplementation. While human RCTs specifically in women over 40 are still emerging, the mechanistic evidence and animal data are directly relevant to this age group. Spermidine cannot restore eggs that have already been lost, but it can help optimize the cellular performance of remaining eggs — which is precisely what matters for women navigating advanced maternal age.
How does spermidine compare to CoQ10 for fertility?
How does spermidine compare to CoQ10 for fertility?
CoQ10 and spermidine are complementary, not competing, interventions. CoQ10 supports mitochondrial electron transport chain function as a direct cofactor — improving the efficiency of existing mitochondria. Spermidine induces mitophagy to clear dysfunctional mitochondria and supports biogenesis of new ones — improving the quality of the mitochondrial population. Together, they address mitochondrial health from different angles and are commonly recommended in combination for fertility optimization.
Is liposomal spermidine safe during pregnancy?
Is liposomal spermidine safe during pregnancy?
Evidence for spermidine supplementation during active pregnancy is limited. The general principle — that endogenous compounds consumed in food are not contraindicated — is reassuring, but the research focus has been on preconception optimization rather than pregnancy support. Discuss continuation or cessation of supplementation with your OB-GYN once pregnancy is confirmed. The preconception optimization period is the most critical window for spermidine’s fertility benefits.
What is the relationship between spermidine and PCOS?
What is the relationship between spermidine and PCOS?
Polycystic ovary syndrome (PCOS) involves insulin resistance, chronic inflammation, and oxidative stress — all of which impair follicular development and egg quality through mechanisms that spermidine directly addresses. Several autophagy pathways are impaired in PCOS granulosa cells, suggesting particular relevance for spermidine in this population. While specific PCOS-focused spermidine research is limited, the mechanistic alignment is strong. Myo-inositol (discussed in Section 9) is the best-studied supplement for PCOS-related fertility improvement and is fully compatible with the AOCP.
Conclusion: Cellular Health Is the New Frontier of Fertility
The science is compelling, the mechanism is precise, and the opportunity is real: spermidine is not a fertility fad — it is a fundamental biological molecule whose age-related decline tracks directly with reproductive aging, and whose restoration activates the cellular repair systems that eggs and sperm need to function at their best.
Across a growing body of research — from the landmark Zhang et al. 2023 Nature Aging study demonstrating oocyte rejuvenation via mitophagy, to multiple independent studies on follicular atresia reduction, granulosa cell protection, porcine oocyte quality preservation, and male spermatogenesis support — a consistent picture has emerged: restoring spermidine levels in aging reproductive tissues activates autophagy, clears mitochondrial damage, reduces oxidative stress, and meaningfully improves the cellular quality of both eggs and sperm.
For the educated, research-driven women aged 30–45 navigating the complex terrain of age-related fertility decline — this is not wishful thinking. It is targeted cellular biology applied to the most important project of your life. For the men aged 30–45 who are ready to take genuine ownership of their 55% stake in this equation — the 74-day spermatogenic cycle is your roadmap to measurable improvement. For couples facing IVF — the 1% cost investment in cellular optimization relative to a single cycle’s expense is rational biological insurance.
Key Takeaways from This Guide: Spermidine activates autophagy and mitophagy — the primary cellular quality control systems for eggs and sperm. Spermidine levels decline by up to 60% with age, correlating with declining fertility. Liposomal spermidine formulations provide substantially better bioavailability than standard oral supplements. The Autophagy-Optimized Conception Protocol (AOCP) provides a 3–6 month framework for comprehensive preconception cellular optimization. Spermidine is safe, well-tolerated, and complementary to both natural conception and IVF medical protocols.
Preconception Window
The preconception window is finite and precious. The decisions made in the months before conception — about nutrition, supplements, lifestyle, and cellular health — shape the biological environment in which new life begins. Spermidine science gives couples the tools to make that environment as healthy as possible. The research supports it. The safety profile allows it. The stakes demand it.
Begin early. Be consistent. Stay informed. And share your journey — including your labs, your outcomes, and your questions — with your healthcare team. Together, evidence-based preconception optimization and skilled medical care represent the most powerful combination available for couples building their families in the 21st century.
Key Research References and Citations
The following peer-reviewed studies and resources are cited throughout this guide. All PubMed and PMC links lead to National Library of Medicine database entries. This list is provided for transparency and to enable independent verification of claims; it is not exhaustive of all research in this field.
- Female Fertility / Oocyte Quality — Landmark Study: Zhang, Y. et al. (2023). Spermidine restores maternal mitophagy and thereby improves oocyte quality and fertility in mice. Nature Aging. https://pubmed.ncbi.nlm.nih.gov
- Follicular Atresia and Ovarian Protection: Multiple authors. Spermidine reduces follicular atresia and oxidative stress in aged mouse ovaries; autophagy protein markers LC3, Beclin-1. https://pmc.ncbi.nlm.nih.gov
- Post-Ovulatory Oocyte Quality (IVF Relevance): Spermidine improved spindle integrity, cortical granule distribution, fertilization rates in aged porcine oocytes. https://pubmed.ncbi.nlm.nih.gov
- Triclosan-Exposed Porcine Oocytes: Spermidine protects against toxin-induced meiotic defects and fertilization impairment via oxidative stress attenuation. https://www.researchsquare.com
- Male Fertility — Testicular Protection: Spermidine rescue of triptolide-induced testicular dysfunction; polyamine restoration, HSP70 upregulation. https://pubmed.ncbi.nlm.nih.gov
- Fasting-Mimicking Diet and Autophagy: Human pilot trial demonstrating FMD-induced autophagic flux improvement, IGF-1 reduction, metabolic benefits. https://pubmed.ncbi.nlm.nih.gov
- Spermidine, Autophagy, and Longevity: Eisenberg, T. et al. Induction of autophagy by spermidine promotes longevity. Nature Cell Biology, 2009. https://pubmed.ncbi.nlm.nih.gov
- Spermidine and Sperm Oxidative Stress: Research documenting spermidine’s reduction of seminal ROS and improvement in sperm motility/morphology parameters. AARUX Pharma nutraceutical research.
- Autophagy in Ovarian Biology: NIH / National Library of Medicine resources on autophagy in folliculogenesis, oocyte maturation, and corpus luteum function. https://www.ncbi.nlm.nih.gov
- Male Attitudes Toward Fertility: NIH research on male reproductive health awareness and preconception health behaviors. https://www.ncbi.nlm.nih.gov
Legal Disclaimer
This article is for informational and educational purposes only and does not constitute medical advice, diagnosis, or treatment. The information presented is based on scientific research available at the time of writing and is subject to change as new evidence emerges. Individual fertility situations vary significantly. Always consult with a qualified healthcare provider, reproductive endocrinologist, or specialist before beginning any supplementation regimen, making changes to existing treatment protocols, or acting on any information in this article. The author and publisher assume no liability for actions taken based on this content.
Pingback: How to Produce the Healthiest Eggs and Sperm for Conception: The Science of Preconception Optimization - progenybrands.com
Pingback: How to Improve Egg Quality After 35 - progenybrands.com
Pingback: Spermidine and Male Factor Infertility - progenybrands.com
Pingback: The Autophagy Fertility Optimized Conception Protocol - progenybrands.com
Pingback: Mitophagy and Egg Quality: Reversing Age-Related Mitochondrial Decline - progenybrands.com
Pingback: The Role of Autophagy Fertility in Preconception - progenybrands.com
Pingback: The Spermidine-Fertility Parallel: How Declining Spermidine Levels Mirror Egg & Sperm Quality Decline [Infographic] - progenybrands.com
Pingback: Liposomal Spermidine vs. Regular Spermidine: The Bioavailability Breakthrough for Preconception Optimization, Egg Quality, and Sperm Health - progenybrands.com
Pingback: Male Attitudes Toward Fertility Supplements – Why 55% of Cases Are Male Yet Underdiscussed - progenybrands.com
Pingback: Fasting-Mimicking Diet for Autophagy: Human Markers and Preconception Results - progenybrands.com
Pingback: Cellular Fertility Optimization: Unlocking Healthier Eggs, Sperm, and Conception at the Cellular Level - progenybrands.com
Pingback: Polyamine Fertility Support: Unlocking Preconception Optimization Through Cellular Renewal and Autophagy - progenybrands.com